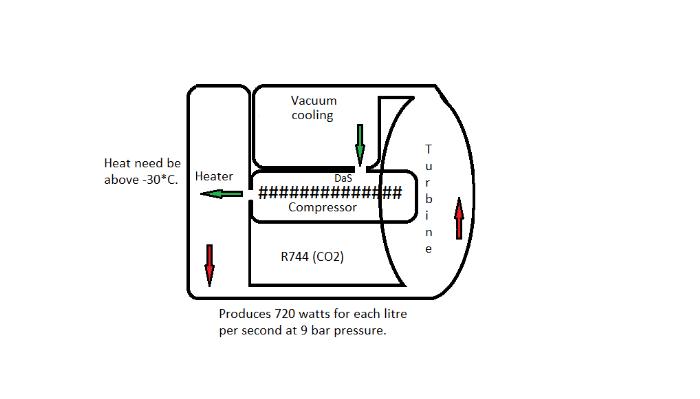
1
1








The colour usually shown by silica gel is due to an indicator, added to see directly when it is dehydrated and when it has absorbed moisture. For many years, cobalt chloride (Cl2Co) has been used. This substance gives the dehydrated gel a strong blue colour and a pale pink colour to the gel having absorbed moisture. Recently, the European Union banned its use because of considering it carcinogen through inhalation. A search for new alternatives led to some iron salts, where the change in colour can be poorly distinguished. At present, the most advisable alternative is methyl violet, which gives the dehydrated gel an orange colour and a green colour to the hydrated gel.
























Marcos Buenijo wrote:A chest freezer that consumes one KWh of electricity each day sees a cooling load of roughly 5000 btu over this period. This would require about a gallon of ammonia.








Paramount Natural Design-Build Architect, Engineering Services, GC, LLC.
















If your dreams don't scare you they aren't big enough




If your dreams don't scare you they aren't big enough

|
This tiny ad is guaranteed to be gluten free.
grow your own garden and build your own home in the gardening gardeners program
https://wheaton-labs.com/bootcamp
|